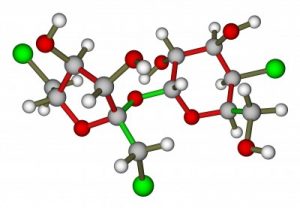
Before I get started, remember that sucralose = Splenda. These two diet sweeteners are the same thing.
Sucralose is marketed as Splenda in America as a “low-calorie” diet sugar. It was created in a UK laboratory to compete with aspartame after NutraSweet’s patent ran out.
My colleague in England shared the sucralose formula with me before its inventors took it out of the public domain.
Basically, sucralose (Splenda) is marketed as a benign sugar with a touch of chlorine, but you might disagree when you read the other side of this equation.
The inventors named it sucralose because it sounds like sucrose (real sugar). They created it by starting with a real sugar molecule. Then they removed 3/4 of that sugar molecule and replaced the space with chlorine.
Using a complex process involving dozens of chemicals that you and I can barely pronounce – let alone consume – I have outlined below the actual process for producing this diet sweetener. I highlighted the “chemicals used” in bold.
Here’s Splenda’s chemical formula: 1,6-dichloro-1, 6-dideoxy-BETA-D-fructofuranosyl-4-chloro-4-deoxy-alpha-D-galactopyranoside.
If you want to read all the details and the research docs, check out my book on Splenda.
How They Make Sucralose

According to the Splenda International Patent A23L001-236 and PEP Review #90-1-4 (July 1991), sucralose is synthesized by this five-step process:
1. sucrose is tritylated with trityl chloride in the presence of dimethylformamide and 4–methylmorpholine and the tritylated sucrose is then acetylated with acetic anhydride,
2. the resulting TRISPA (6,1′,6′-tri-O-trityl-penta-O-acetylsucrose) is chlorinated with hydrogen chloride in the presence of toluene,
3. the resulting 4-PAS (sucrose 2,3,4,3′,4′-pentaacetate) is heated in the presence of methyl isobutyl ketone and acetic acid,
4. the resulting 6-PAS (sucrose 2,3,6,3′,4′-pentaacetate) is chlorinated with thionyl chloride in the presence of toluene and benzyltriethylammonium chloride, and
5. the resulting TOSPA (sucralose pentaacetate) is treated with methanol (wood alcohol, a poison) in the presence of sodium methoxide to produce sucralose.
And they brag that it’s just like sugar.

The Hidden Chemicals in Splenda
The chemicals used to synthesize sucralose in the five-step process (above) are more than just a simple benign sugar with a touch of chlorine. The chemicals used to make sucralose (Splenda) are: (in alphabetical order)
- Acetone
- Acetic acid
- Acetyl alcohol
- Acetic anhydride
- Ammonium chloride
- Benzene
- Chlorinated sulfates
- Ethyl alcohol
- Isobutyl ketones
- Formaldehyde
- Hydrogen chloride
- Lithium chloride
- Methanol
- Sodium methoxide
- Sulfuryl chloride
- Trityl chloride
- Toluene
- Thionyl chloride

There’s no “free ticker” to eating the diet sweeteners.
I tell people that the side-effects will be the same as side-effects to chlorine and these other chemicals.
It’s time to admit that there is no free ticket to eating all the sugar-free products you desire without paying the high price of harming your body in the long run. Laboratory chemicals are not the sugar-free solution to health and wellness.
It certainly doesn’t sound very appetizing to me.
___________________________
If you want to learn more about disease prevention and a healthy lifestyle, contact me at janethull.com. Remember that you are never alone when you are looking for good health!
Gain access to all of my online programs, ongoing support, monthly Q&A, and more. I look forward to supporting you on your journey to alternative health and wellness.
_____________
Disclaimer: This article is for informational purposes only, and is educational in nature. The FDA may not have evaluated some of the statements. This article is not intended to diagnose, treat, cure, or prevent any disease. Please discuss with your own, qualified health care provider before adding supplements or making any changes to your dietary program.
Before taking vitamins, consult your doctor; pre-existing medical conditions or medications you are taking can affect how your body responds to multivitamins.
You have our permission to reprint this article if you attribute us with a live back-link to this article and the youtube links. https://janethull.com/